Cardiology
Access innovative heart and vascular treatments through leading cardiovascular clinical research studies.

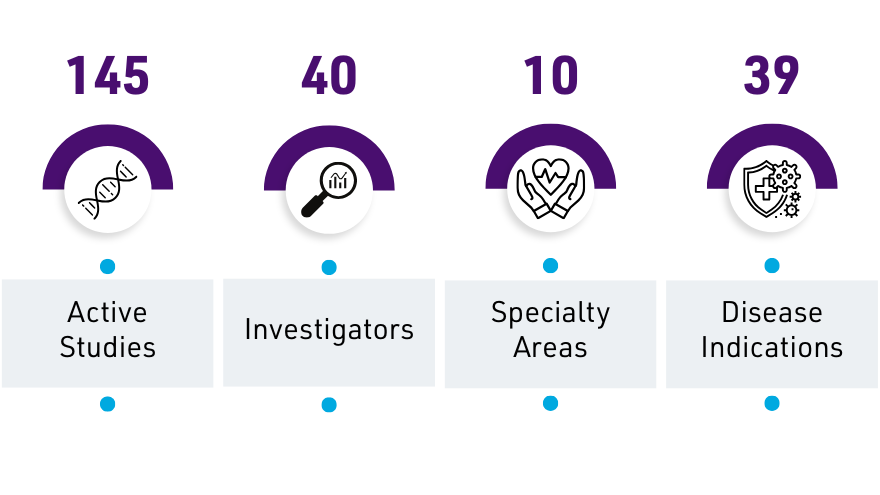
The Renown Health Office of Clinical Research (OCR) connects patients with local clinical trials and research studies in Northern Nevada. Browse clinical trials by specialty or visit our Clinical Trials Directory.